Adhesive Drying & Curing - What's the difference
Published: 08 February 2021
Updated: 20 January 2023
‘Drying’ and ‘Curing’ are two words that we regularly see people confuse when referring to the use of roofing adhesives. From a chemistry point of view, the two have very different meanings.

What does 'Drying' mean?
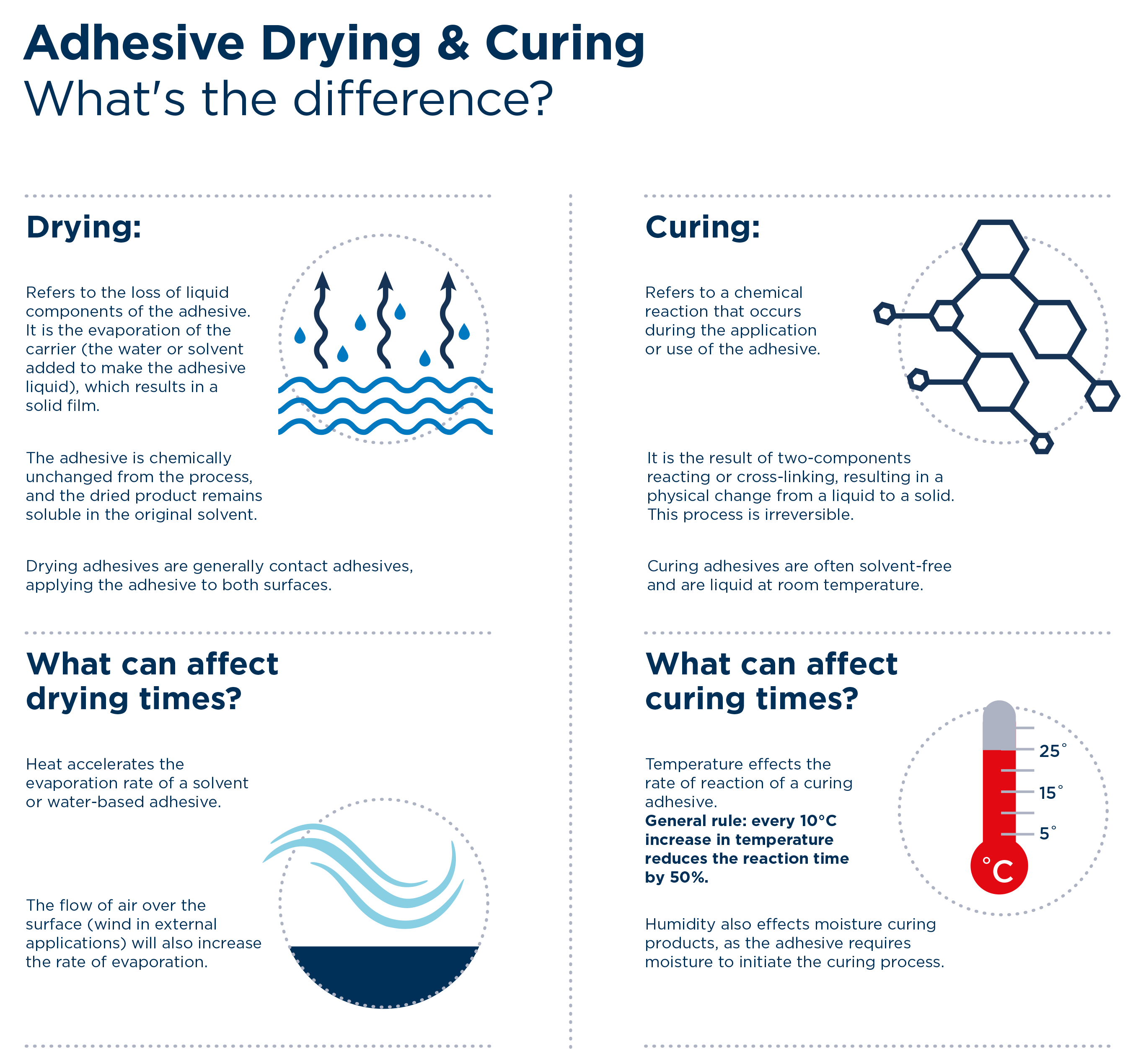
Drying means the loss of liquid components of the adhesive. It is the evaporation of the carrier (the water or solvent added to make the adhesive liquid), which results in a solid film. The adhesive is chemically unchanged from the process, and the dried product remains soluble in the original solvent.
This terminology generally refers to solvent-based contact and water-based adhesives.
What does ‘Curing’ mean?
Curing refers to a chemical reaction that occurs during the application or use of the adhesive. It is the result of two-components reacting or cross-linking, resulting in a physical change from a liquid to a solid. This process is irreversible.
This terminology refers to moisture-curing polyurethanes, two-component polyurethanes and epoxy-based adhesives.
Why do I need to know the difference?
It is important to understand the difference between drying and curing for product selection.
Adhesives that cure have higher temperature and water-resistance compared to products that dry. So ensuring you have the correct adhesive type required for the specific application is vitally important.
Drying adhesives are generally contact adhesives, applying the adhesive to both surfaces. These adhesives have varying tack times. The rate of evaporation of the solvent affects the tack times.
Curing adhesives are often solvent-free and are liquid at room temperature. These types of adhesives have varying open times and cure times which are determined by the rate of reaction of the adhesive system.
What can affect adhesive drying and curing times?
Temperature and humidity can drastically affect a roofing adhesive’s drying or curing time.
Heat accelerates the evaporation rate of a solvent or water-based adhesive. Also, the flow of air over the surface (wind in external applications) will dramatically increase the rate of evaporation of solvents and water from an adhesive film.
Temperature effects the rate of reaction of a curing adhesive. A general rule is every 10°C increase in temperature reduces the reaction time by 50%. Humidity also effects moisture curing products, as the adhesive requires moisture to initiate the curing process. Often a water over-mist is applied to a moisture cuing polyurethane to ensure the adhesive cures correctly.
Our infographic summaries the key points:
Sign up to our newsletter to get our blogs delivered straight to your inbox
This form is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.









